Drug discovery has always been one of the most expensive and failure-prone endeavors in modern science. It costs more than $2 billion on average to bring a single drug to market, the process takes ten to fifteen years from initial discovery to pharmacy shelf, and roughly nine out of ten drug candidates that enter human trials will fail before reaching patients.
AI companies have been promising to change those odds for years. In 2025, for the first time, there was real clinical evidence they might be right. A drug discovered and designed entirely by artificial intelligence completed a Phase IIa trial with results published in Nature Medicine. As of early 2026, more than 173 AI-discovered drug programs are in clinical development, with 15 to 20 expected to enter pivotal Phase III trials during the year.
The number that the industry is watching most closely is not 173. It is 15. Phase III is where most pharmaceutical failures happen, and it is where AI's early advantages in speed and hit rates will either translate into clinical success or fail to matter at all.
Why 2026 Is Different
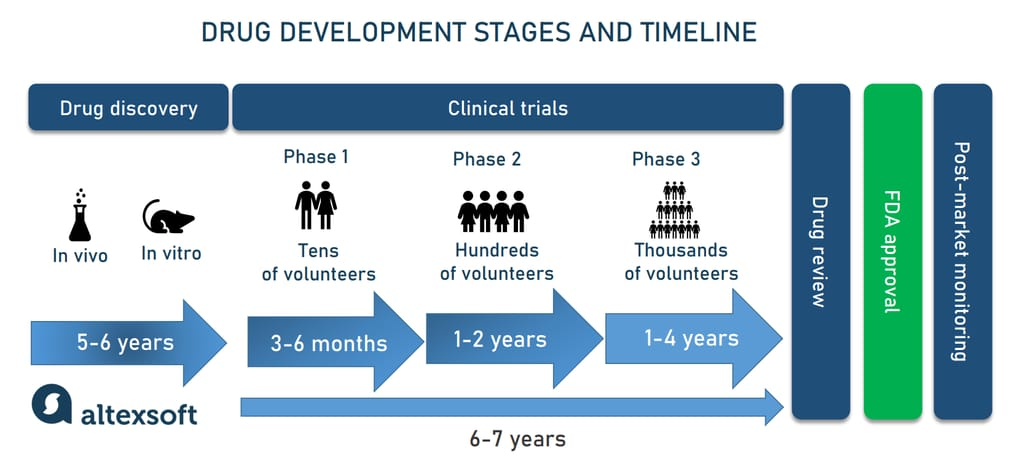
Most AI drug discovery coverage to date has focused on what happens before clinical trials: faster target identification, cheaper molecular design, higher hit rates in preclinical screening. Those advantages are real and well-documented. AI-discovered compounds achieve Phase I success rates of roughly 80 to 90 percent, significantly higher than the 52 percent historical average for traditional methods.
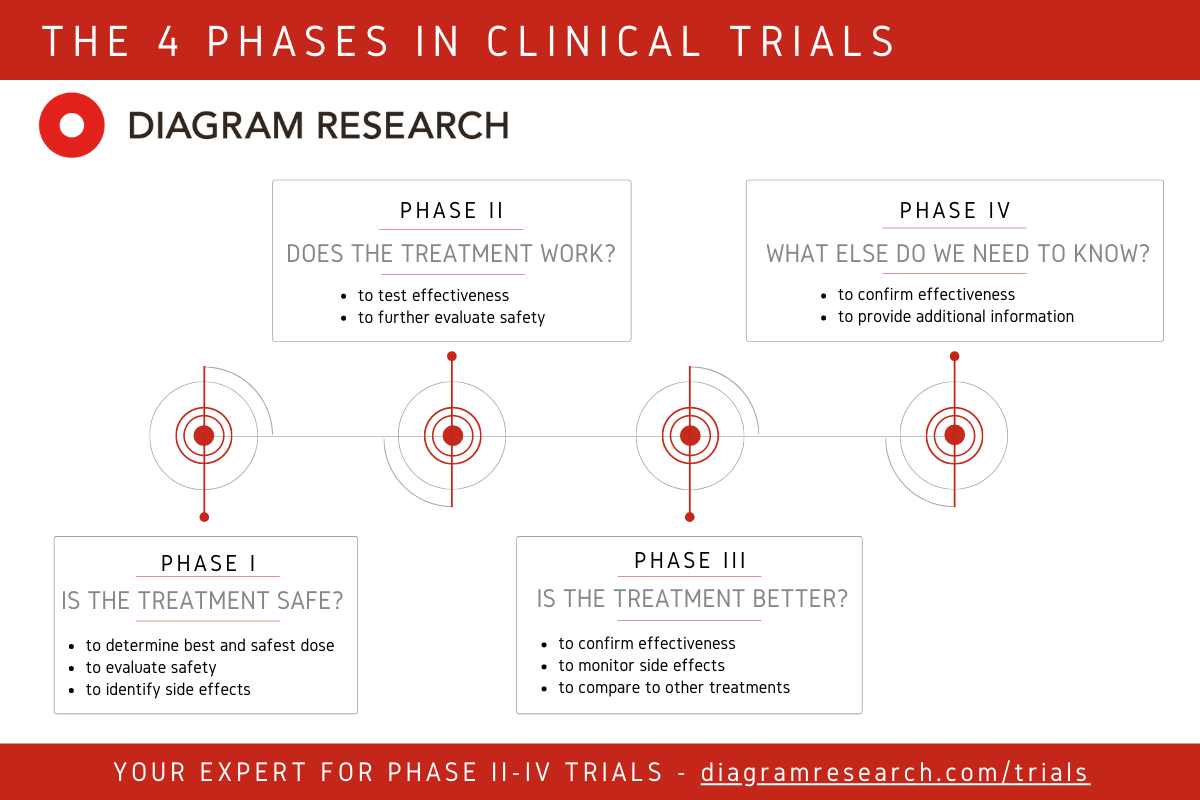
Phase I tests whether a drug is safe and tolerable. Phase II begins exploring efficacy in a defined patient population. Phase III is the definitive test: large-scale, randomized, controlled, often running for years, and requiring proof of meaningful clinical benefit that satisfies regulators.
No fully AI-designed drug has completed all trial phases and received regulatory approval. That milestone is projected to arrive in 2026 or 2027, with roughly 60 percent probability by independent analysts. The Phase III readouts arriving in 2026 are the first serious evidence base for whether AI's early clinical advantages hold through to the trials that actually determine approval.
As one analysis framed it: "AI has made the early part of drug development faster and cheaper. Whether it can make the clinical part more successful is the question 2026 is beginning to answer."
The Drug at the Center: Rentosertib and Insilico Medicine
The most closely watched compound in this space is rentosertib, formerly designated ISM001-055, developed by Insilico Medicine. It holds a specific distinction that makes it the industry's most meaningful data point: it is the first drug in which both the disease target and the molecular compound were identified and designed using generative AI, with no human hypothesis guiding either step.
The target identification and the molecule are both AI-originated, making rentosertib a true end-to-end test of what the technology can do.
The Disease: Why IPF Matters as a Test Case
Rentosertib targets idiopathic pulmonary fibrosis, a chronic progressive lung disease characterized by irreversible scarring of lung tissue. IPF affects roughly five million people worldwide, has few effective treatments, and typically kills patients within two to five years of diagnosis. Because patients must take any treatment continuously for the rest of their lives, the drug faces stringent safety requirements alongside its efficacy bar.
Insilico chose IPF specifically because it is a hard problem. The entire biological rationale for why a particular protein, TNIK, was implicated in fibrosis was something the AI identified from multi-omics data, not a mechanism the research team already suspected.
How It Was Built
Insilico's target discovery platform, PandaOmics, was trained on omics and clinical datasets related to tissue fibrosis and used deep feature synthesis and causality inference to identify TNIK as a novel target. The company's Chemistry42 platform then generated 78,000 virtual TNIK inhibitors, filtered through multi-objective optimization covering potency, selectivity, and synthesizability, and ranked 60 top candidates for physical synthesis.
The hit rate was 16.7 percent. Traditional high-throughput screening produces hit rates of around 0.1 percent.
From novel target identification to Phase I human trials took 30 months. The comparable timeline for traditional drug discovery is six to eight years, at a fraction of the cost.
What the Phase IIa Results Show
Results from the Phase IIa trial were published in Nature Medicine in June 2025. In the primary efficacy analysis, patients receiving the highest dose of 60 mg once daily showed a mean improvement in forced vital capacity of 98.4 milliliters. The placebo group showed a mean decline of 62.3 milliliters over the same period. That is a difference of more than 160 milliliters in a disease where slowing decline, rather than reversing it, has historically been the treatment ceiling.
The results are promising, but they are not yet proof of approval-level efficacy. Phase IIa trials are smaller, shorter, and less statistically demanding than Phase III. Insilico is pursuing regulatory discussions for a pivotal Phase III trial as of early 2026, with a separate Phase IIa trial in US patients actively ongoing.
Schrödinger and Zasocitinib: A Different Approach Already in Phase III
While rentosertib is the most discussed AI-designed compound, it is not the furthest along in clinical development. That distinction belongs to zasocitinib, also known as TAK-279, developed through a collaboration between Schrödinger and Nimbus Therapeutics and subsequently acquired by Takeda. Zasocitinib is already in Phase III trials for psoriasis and other inflammatory conditions.
Schrödinger's approach is technically distinct from Insilico's generative chemistry. Rather than using machine learning to generate novel molecular structures from scratch, Schrödinger combines quantum mechanical simulations with machine learning to predict how molecules interact with protein targets at the atomic level. The system models the physics of molecular binding with a precision that data-driven generative approaches cannot always achieve, particularly for targets where three-dimensional binding geometry is critical.
The zasocitinib Phase III readouts expected in 2026 will be the first large-scale clinical test of a physics-based AI design approach. Schrödinger's role was in predicting the precise molecular interactions that made zasocitinib a stronger candidate than alternatives, compressing optimization work that would otherwise have taken years. This is better understood as AI-enhanced physics than as an independently AI-originated compound in the same sense as rentosertib.
Recursion-Exscientia: The Merged Platform and What Is Coming in 2026
The merger of Recursion Pharmaceuticals and Exscientia, completed in July 2025, created the largest combined AI drug discovery entity by pipeline breadth and proprietary data. The merged company integrates Recursion's high-throughput biological imaging, which runs approximately 2.2 million experiments per week across 50 human cell types, with Exscientia's precision molecular design tools and more than 60 petabytes of proprietary data.
The combined pipeline includes more than 10 clinical and preclinical programs. The company has received over $450 million in partnership payments to date and holds more than $20 billion in potential future milestones.
Two programs are generating the most near-term data in 2026.
The merged entity also carries the weight of a prominent failure. REC-994, the lead AI-discovered compound from Recursion's pre-merger pipeline targeting cerebral cavernous malformation, was discontinued in May 2025 after long-term data did not confirm the efficacy trends seen in earlier studies. It is the most high-profile AI drug discovery failure to date, and it matters as a counterweight to narratives that treat AI as a guaranteed improvement on traditional pharmaceutical development.
The Technical Approaches Behind the Numbers
The term "AI drug discovery" covers a range of meaningfully different technical methods, and the distinctions matter for understanding what each platform can and cannot do.
- Generative chemistry is what Insilico Medicine uses. The platform learns the relationship between molecular structure and biological activity across millions of known compounds, then designs new candidates optimized for multiple properties simultaneously. This approach can explore chemical space that human chemists have never visited, and it produces synthesizable molecules rather than theoretically interesting ones.
- Physics-based AI is Schrödinger's approach. Quantum mechanical simulations predict molecular behavior at the atomic level, with machine learning accelerating the computational work. This is extremely accurate for targets where three-dimensional binding geometry is well characterized, but computationally intensive and less suited to exploratory searches across novel chemical space.
- Phenomics and biological imaging is Recursion's foundational method. Rather than starting with a molecular hypothesis, the platform perturbs cells with thousands of genetic and chemical conditions, captures high-resolution images of cellular responses, and uses computer vision to detect patterns that reveal disease biology and potential interventions. The approach can surface candidates for diseases where the underlying biology is poorly understood.
- Structure-based design using AlphaFold is the approach associated with Isomorphic Labs, Google DeepMind's drug discovery subsidiary. AlphaFold's protein structure predictions enable structure-based design for proteins whose three-dimensional structure was previously unknown or expensive to determine experimentally. Isomorphic Labs has major research partnerships with Eli Lilly and Novartis, though no Isomorphic-originated compounds have yet entered clinical trials.
Each of these approaches has different strengths, different failure modes, and different evidence bases. Treating them as equivalent under the "AI drug discovery" label obscures more than it reveals.
The Failures That Matter
The honest accounting of AI drug discovery in 2025 and early 2026 includes failures that are underreported in coverage focused on the technology's promise.
In 2023, AI-designed compounds missed primary endpoints in clinical trials for atopic dermatitis, schizophrenia, and cancer. These were high-profile tests of what was then a newer generation of AI-designed therapeutics, and they failed in the same ways traditional drugs fail: the clinical benefit was insufficient at the doses tolerated by patients.
REC-994's discontinuation in May 2025 is the most significant single failure in the field's recent history. Recursion's platform identified it as a candidate for cerebral cavernous malformation, a rare vascular condition with no approved treatments. Early clinical data was encouraging enough to advance the program, but long-term data did not confirm the earlier trends, and the program was stopped.
Exscientia's first-ever AI-designed clinical compound, DSP-1181, developed with Sumitomo Pharma for obsessive-compulsive disorder, was discontinued after Phase I. In AI's defense, approximately 90 percent of all compounds tested in Phase I fail regardless of how they were discovered.
These failures do not invalidate the AI drug discovery thesis. They mean that the field's success rates, while better than historical averages in Phase I, are not immune to the fundamental biological risks that make drug development hard. The question is not whether AI can eliminate failure. It is whether AI shifts the probability distribution of success enough to change the economics of pharmaceutical R&D over the long term.
What Regulators Are Actually Doing
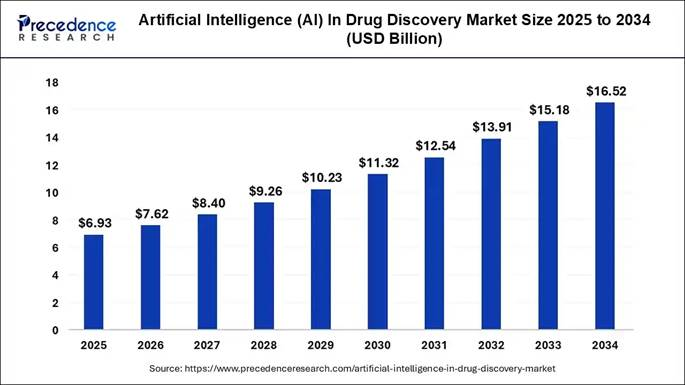
The regulatory environment for AI in drug development has been developing in parallel with the clinical pipeline, and 2026 is a significant year for regulatory frameworks as well as clinical readouts.
In January 2025, the FDA published draft guidance titled "Considerations for the Use of Artificial Intelligence to Support Regulatory Decision Making for Drug and Biological Products." It was the first comprehensive regulatory framework specifically addressing AI throughout the drug development lifecycle, establishing a risk-based credibility assessment approach. Companies using AI to support regulatory submissions must demonstrate that their models are appropriate for the specific intended use and that outputs have been validated against relevant benchmarks.
Final guidance is expected in the second quarter of 2026. When it arrives, it will define the documentation requirements, validation standards, and submission formats that companies must follow when AI has contributed to any part of their development process. Every company with compounds in Phase III will be affected, because the data they submit will need to meet those standards.
In January 2026, the FDA and the European Medicines Agency jointly released non-binding guiding principles for good AI practice in drug development. This joint release signals regulatory coordination between the two agencies that govern most of the world's pharmaceutical approvals, and it suggests that whatever framework the FDA finalizes will have meaningful alignment with EMA expectations.
What the First Approval Would Actually Mean
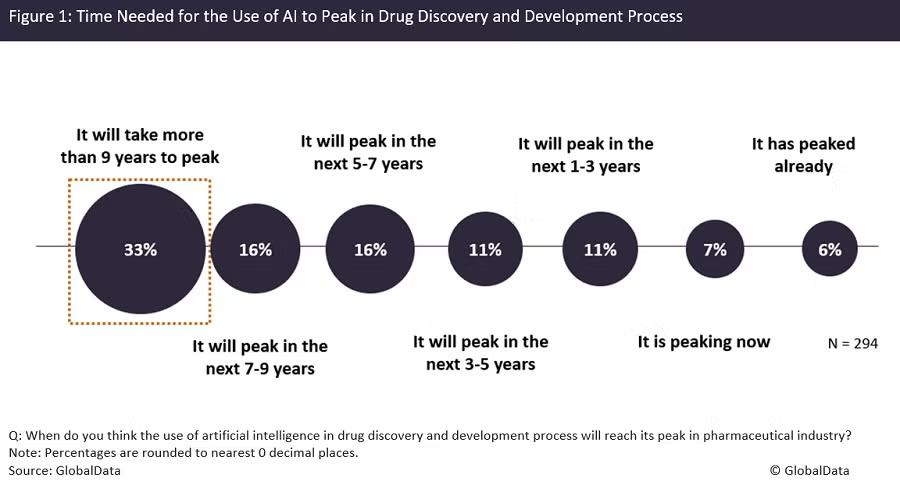
The first regulatory approval of a fully AI-designed drug, projected for 2026 or 2027, would be a genuine landmark. It would mean that a compound identified by AI as a target, designed by AI as a molecule, and optimized by AI for safety and efficacy successfully passed the full clinical and regulatory gauntlet that has historically filtered out 90 percent of all drug candidates.
The significance would extend beyond the specific drug and disease. It would validate the end-to-end AI drug design process in a way that no amount of Phase I or Phase II data can. It would establish a regulatory precedent for how AI-originated compounds are reviewed and change the investment calculus for the entire sector, where billions of dollars in capital have been committed to a thesis that has not yet produced a single approved drug.
A single approval would not, however, prove that AI drug discovery is reliably superior to traditional methods. It would prove that one AI-designed drug worked well enough in one disease to satisfy one regulatory standard. The broader validation will come from the pattern of Phase III results across the 15 to 20 pivotal trials arriving in 2026 and beyond.
Wrap up
The AI drug discovery field has spent years producing evidence that it can make the early parts of pharmaceutical R&D faster and cheaper. Hit rates are up. Time to preclinical candidate selection is down. Phase I success rates are higher than historical averages. The speed advantage demonstrated by rentosertib, from target to Phase I in 30 months versus the traditional six to eight years, is real.
What the field has not yet proven is that these early advantages translate into clinical success at the scale that regulators and patients require. Phase III is the hardest test drug discovery has always faced, and AI has not yet run it at meaningful scale.
The promises made by AI drug discovery companies have been large and specific: faster timelines, higher success rates, more drugs for diseases that traditional approaches have failed to address. Some of those promises have early supporting evidence. None of them are proven. 2026 is the year the evidence starts arriving in the form that actually matters: large-scale, controlled, randomized trials with regulatory-grade outcomes. Whether the promises were real is a question that will begin to have a real answer.
Frequently Asked Questions
How many AI-discovered drugs are currently in clinical trials?
As of early 2026, more than 173 AI-discovered drug programs are in clinical development. The breakdown is roughly 94 programs in Phase I, 56 in Phase II, and 15 in Phase III, with 15 to 20 additional programs expected to enter pivotal Phase III trials during 2026. No fully AI-designed drug has yet received regulatory approval, though the first approval is projected for 2026 or 2027 with approximately 60 percent probability by independent analysts.
What is rentosertib and why is it important?
Rentosertib, formerly designated ISM001-055, is developed by Insilico Medicine and is the first drug in which both the disease target and the molecular compound were identified and designed by generative AI. It targets idiopathic pulmonary fibrosis, a progressive lung disease with few effective treatments. Phase IIa results published in Nature Medicine in June 2025 showed patients receiving the highest dose had a mean improvement in lung function of 98.4 milliliters compared to a decline of 62.3 milliliters in the placebo group. Insilico is pursuing regulatory discussions for a pivotal Phase III trial as of early 2026.
Do AI-designed drugs have better success rates than traditional drugs?
Early data suggests they do in Phase I, with success rates of 80 to 90 percent compared to the roughly 52 percent historical average for traditional methods. Phase II rates also appear higher. However, Phase III results, where most pharmaceutical failures occur, remain limited. The 2026 pivotal readouts are the first large-scale test of whether AI's early advantage translates to better ultimate outcomes. Failures in 2023 and the discontinuation of Recursion's REC-994 in 2025 demonstrate that AI has not eliminated clinical failure.
What is the difference between Insilico, Recursion, and Schrödinger's AI approaches?
They use fundamentally different methods. Insilico uses generative chemistry, designing novel molecular structures optimized for multiple properties simultaneously with no predetermined template. Schrödinger uses physics-based AI, combining quantum mechanical simulations with machine learning to predict molecular interactions at the atomic level. Recursion uses phenomics, perturbing cells under thousands of conditions and using computer vision to identify disease patterns without starting from a molecular hypothesis. Each approach has different strengths and works better in different target classes.
What happened when AI drug discovery has failed?
There have been several notable failures. In 2023, AI-designed compounds missed primary endpoints in trials for atopic dermatitis, schizophrenia, and cancer. In May 2025, Recursion discontinued its lead AI-discovered candidate REC-994 for cerebral cavernous malformation after long-term data did not confirm earlier efficacy trends. Exscientia's first clinical compound was discontinued after Phase I. These failures reflect that AI can improve early-stage odds but cannot eliminate the biological risks that make Phase II and III hard.
What is the FDA doing about AI in drug development?
The FDA published its first comprehensive draft guidance on AI in drug development in January 2025, establishing a risk-based credibility assessment framework for companies using AI to support regulatory decision-making. Final guidance is expected in the second quarter of 2026. In January 2026, the FDA and the European Medicines Agency jointly released non-binding guiding principles for good AI practice in drug development, signaling coordination between the two agencies governing most of the world's pharmaceutical approvals.
What would the first approved AI-designed drug mean for the industry?
The first regulatory approval of a fully AI-designed drug would validate the end-to-end AI drug design process in a way that no Phase I or Phase II data can achieve. It would establish a regulatory precedent for how AI-originated compounds are reviewed and significantly change the investment calculus for the entire sector. However, it would not prove that AI drug discovery is reliably superior to traditional methods: it would prove that one AI-designed drug worked well enough in one disease. The broader validation will come from the pattern of results across multiple pivotal trials over the coming years.
Related Articles